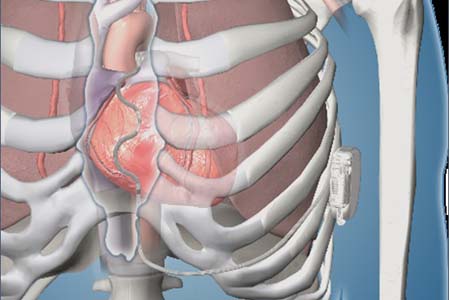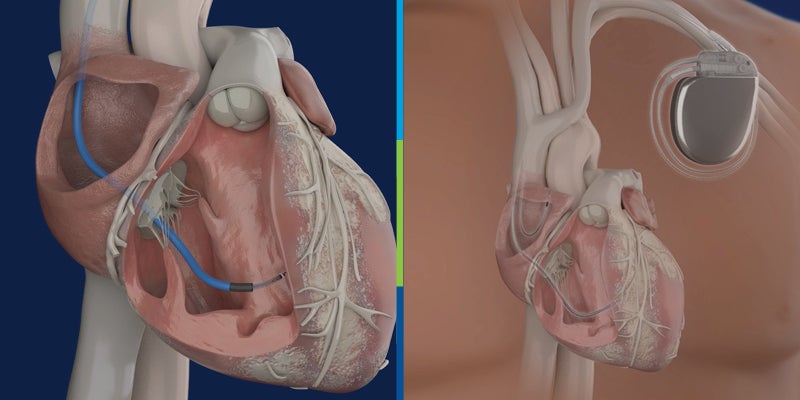
Lehigh Valley Heart and Vascular Institute Participating in World-Wide Implantable Cardiac Defibrillator Study

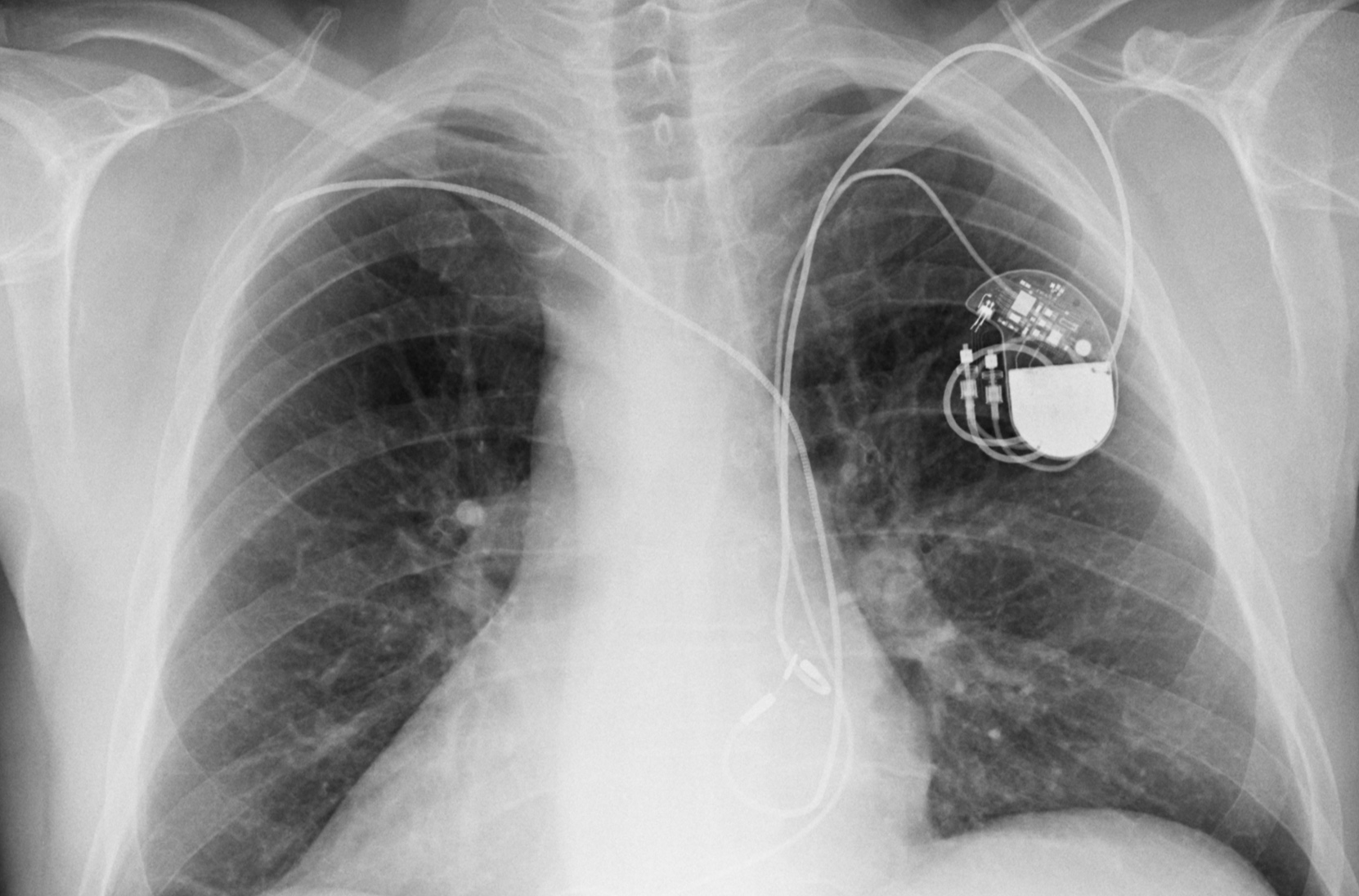
First-in-Human Chronic Implant Experience of the Substernal Extravascular Implantable Cardioverter-Defibrillator - ScienceDirect
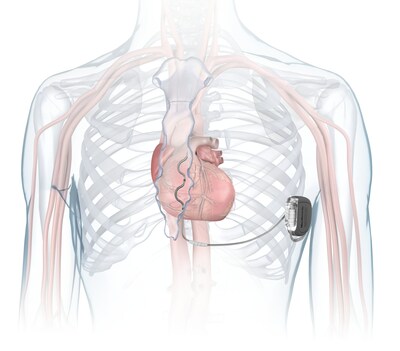
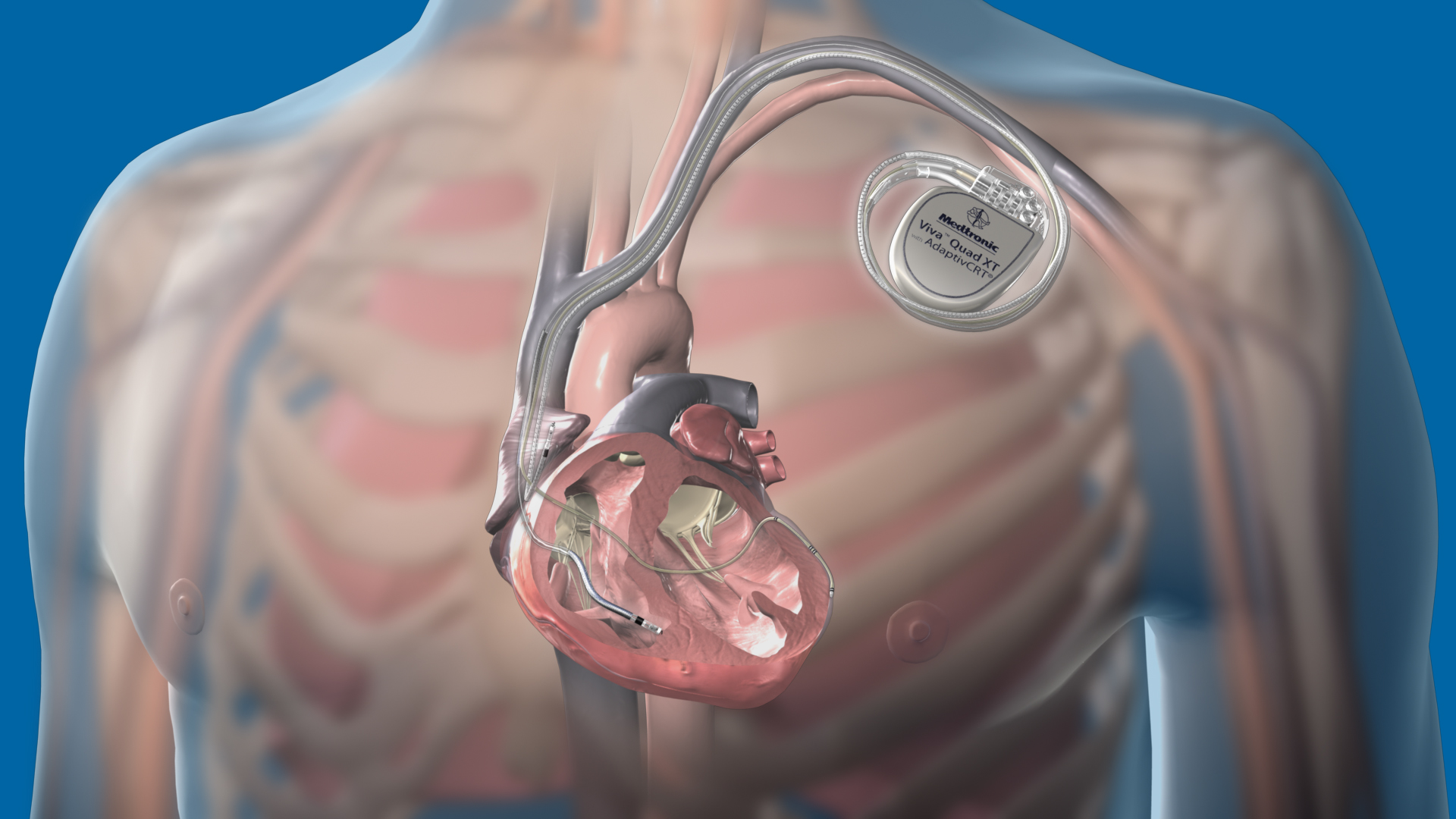
Medtronic receives FDA approval for extravascular defibrillator to treat abnormal heart rhythms, sudden cardiac arrest - Oct 23, 2023

Medtronic Cardiac on X: "See new EV ICD data presented by @Boersma_Lucas at #ESC2023. See results here: https://t.co/XLVRtDRXXA #EPeeps https://t.co/rqQDhTVmJe" / X

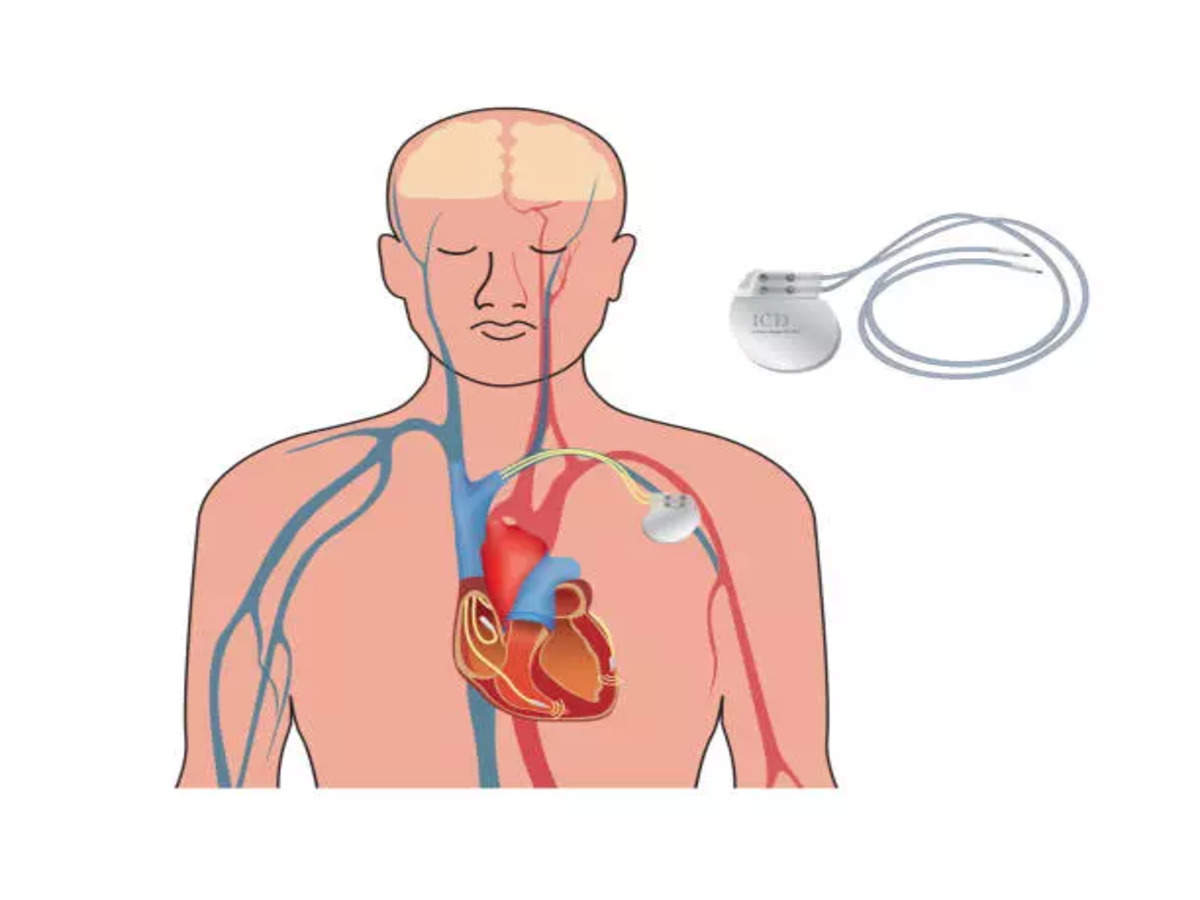
Medtronic Extravascular ICD meets global pivotal clinical trial's safety and effectiveness endpoints | DAIC

Medtronic receives FDA approval for extravascular defibrillator to treat abnormal heart rhythms, sudden cardiac arrest - Oct 23, 2023